Your pharma laboratory is on the front line of manufacturing new medications and driving the development of medical technology. But how can you tone your performance up even further?
Simple. Follow the Good Laboratory Practices (GLP) defined by the appropriate regulatory bodies in your country.
In the US, the Food and Drug Administration (FDA) sets and enforces GLP standards developing, testing, studying and certifying pharmaceuticals and medical devices. Further, the Environmental Protection Agency (EPA) sets GLP standards for the development, testing and safety of pesticides, agrochemicals and food derivatives. In Europe, these regulations are overseen by the European Medicines Agency (EMA) and European National Accreditation Agency (ENAC) respectively.
Worldwide, national and regional institutions take their cues from these organizations to facilitate the acceptance of safety & efficacy standards for new and existing pharmaceutical and food products.
These standards involve analysis of the facilities, equipment, personnel, standard operating procedures (SOPs), testing and reporting systems, QA and QC processes, level of independent monitoring, data collection procedures, and compliance.
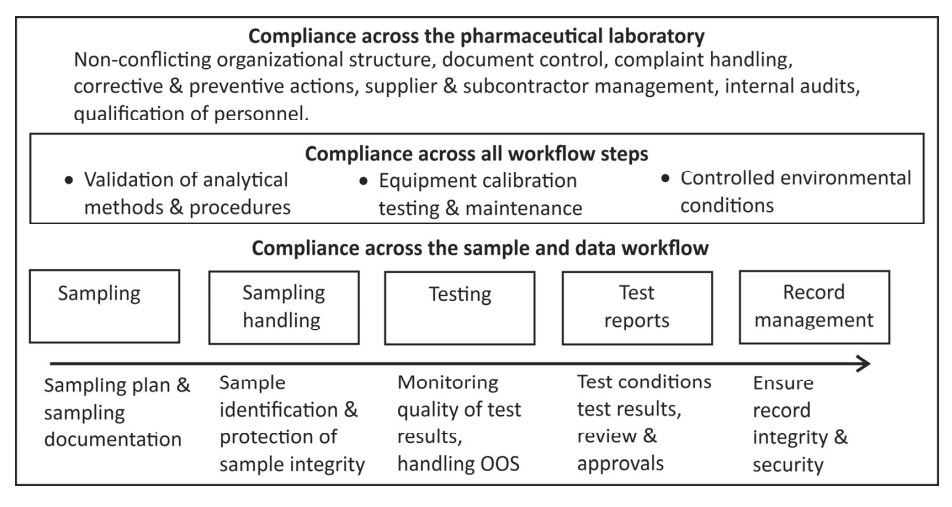
Take a look at some of these best practices for pharma labs, and make sure you and your team are performing at your very best.
Maintain a Log
In today’s largely digitized world, a log can come in a number of forms. However, the crucial element here is to have some kind of record of what you are doing, why you are doing it, how you are doing it, and what the outcomes are, each and every day.
Make this log searchable to increase accountability among you and your team and to create an archive of your processes.
Build upon Basic Procedures
Pharma laboratory work often entails complex calculations, experiments, and processes. These processes may be difficult to get right, which means you have to spend a lot of time planning and honing the way you go about executing them.
But what about the basics? These are things you do every day, and they need to be dialed in for you and your team. You can’t afford to be making mistakes here, so see to it that your training is up to scratch and your personnel are giving their full attention even to the basic tasks.
Test Sharpness and Proficiency
We are all human, so we sometimes take our eyes off the ball. It’s natural, and nothing to be worried about, as long as you are taking steps to make sure it doesn’t affect your laboratory practices.
Along with the training mentioned above, it is important that you conduct regular tests of your personnel’s sharpness and proficiency. You don’t have to put pressure on your staff – simply analyze their performance and offer guidance in a friendly and informal way, and provide additional support where necessary.
Adopt Evidence-Based Processes
Why have you chosen to conduct a certain practice in your laboratory?
Why have you decided to use a certain process or procedure?
If the answer is “because we ran a few tests and decided that Practice/Process/Procedure A was the most effective option,” then this is good.
If the answer is “because we’ve always done that and don’t know any different,” this is not so good.
Basically, everything you do needs to be based on evidence and reason. If not, how can you be sure that you are taking the most effective action?
Do your research into the pharma industry, stay up to date with the latest findings, and test best practices to find out which ones work for you.
Double-Check Hazardous Material Storage and Handling Equipment
If you work in a pharma laboratory, chances are you deal with a lot of hazardous materials. Make sure you have the equipment in place to deal with these materials. For example, deploy secondary containment measures and spill decks to prevent large-scale contamination in the event of a spill.
Retain Data Records
What do you do with your data after you have completed a process? Up until fairly recently, data storage was difficult and expensive, especially in the long term. However, in recent years, cloud structures and other storage concepts have made this far easier.
Now, there is no excuse not to keep your data, and to refer back to this data as and when required.
Double Up on Testing
Whether you are conducting tests on a newly developed medication or on your lab’s wastewater, make sure these tests are run multiple times. As many times as is cost-effective for you and your lab team. One result can easily be an anomaly and can leave you with a skewed set of data. Two results are far less likely to be anomalous, while three sets of results are even more watertight.
Adopting this approach to testing will make your lab’s results all the more reliable going forward.
Updated: 07/10/2024
About Author

Neel Daphtary
Neel Daphtary is the President of Chemtech International. He oversees sales, distribution and business development. He excels at helping pharmaceutical and manufacturing firms find the right processes and environmental solutions. Neel is an active member of Global Philadelphia, an organization committed to community development in PA.






Have Any Questions Or Comments?
Call Us For Help!
888-709-8070